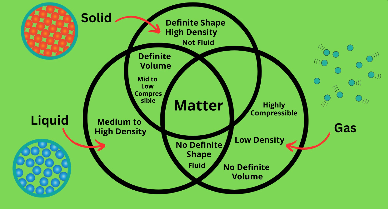
Let's take a look at the similarities and differences between solids, liquids, and gases.
What do they all three have in common?
Solids, liquids, and gases are all three a type of matter, which means that they all have a mass and they all take up space.
Shape
Solids have a definite shape, liquids do not have a definite shape and they take on the shape of the container. Gases also do not have a definite shape.
Volume
Solids have a definite volume, liquids also has a definite volume, however gases do not have a definite volume.
Density
Density is mass divided by volume. A solid has high density, a liquid can have medium to high density and then a gas has a very low density.
Solids and liquids are slightly compressible and gases are highly compressible.
Solids are not considered fluid but gases and liquids are considered fluid.
The Venn Diagram below summarizes the similarities and differences between solids, liquids, and gases.


.png)
0 comments:
Post a Comment